2.1 The nature of matter
|
textbook
|
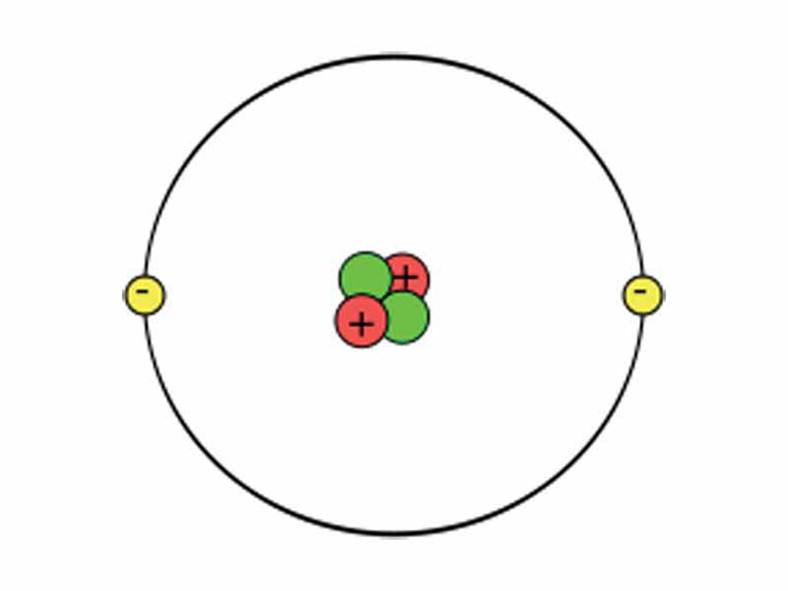
Atoms is a basic unit of matter, incredible small and unable to be cut. Atoms are neutral.
Element is a pure substance that consists entirely of one type of atom Isotopes is atoms of the same elements that differ in the number of neutrons that they contain Chemical compound is a substance formed by the chemical combination of two or more elements in difinite proportions. Chemical Bond is the atoms in compounds are held together by chemical bonds. Two main types of chemical bonds are ionic bonds and covalent bonds. |
2.2 The properties of water
|
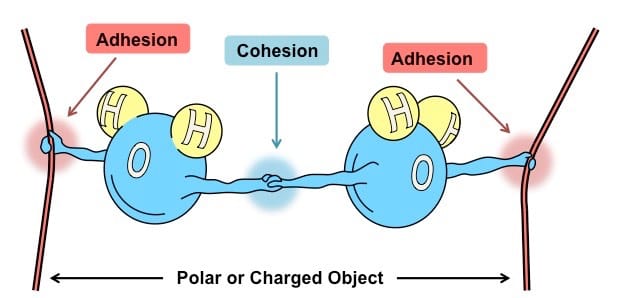
Polar molecule is a molecule in which the charges are nevenly distributed.
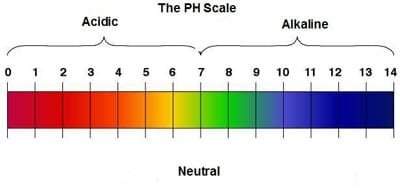
A water molecule is polar because there is an uneven distribution of electrons between the oxygen and hydrogen atoms. Cohesion is an attraction between molecules of the same substance. Adhesion is an attraction between molecules of different substance. Solution and suspensions are two types of mixture that can be made of water. Acid is any compound that forms H+ ions in solution. Acidic solutions contain higher concentrations of H+ ions than pure water and have pH values below 7. Base is a compound that produces hydroxide ions (OH-) in solution. Basic, or alkaline, solutions contain lower concentration of H+ ions than pure water and have pH values below 7. |
http://ib.bioninja.com.au/standard-level/topic-2-molecular-biology/22-water/cohesive-and-adhesive-prope.html
https://medictests.com/acid-base-balance/
|
2.3 Carbon compounds
|
https://dlc.dcccd.edu/biology1-3/carbohydrates
http://anahaacksbiologyportfolio.weebly.com/dna-and-rna.html
|
Macromolecules are formed by a process called polymerization, in which large compounds ( polymers) are built by joining smaller ones ( monomers) together.
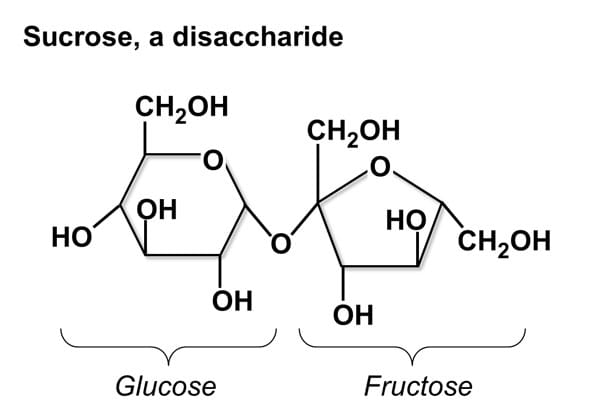
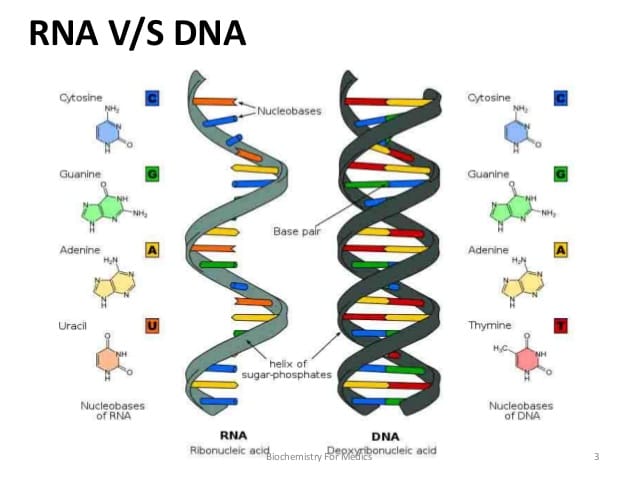
Four groups of organic compounds found in living things are carbohydrates, lipids, nucleic acids and proteins. Carbohydrates are compounds made up of carbon, hydrogen, and oxygen atoms, usually in ratio 1:2:1. Living things use carbohydrate as their main source of energy. Plants and some animals also use carbohydrates for structural purposes. Monosaecharides are single sugar molecules. Polysaccharides are the large macromolecules formed from monosaccharides. Lipids are a large and varied group of biological molecules that are generally not soluble in water, mostly from carbon and hydrogen atoms. Lipids can be used to store energy, some lipids are important parts of biological membranes and waterproof coverings. Nucleic Acids aremacromolecules containing hydrogen, oxygen, nitrogen, carbon, and phosphorus. Nucleotides consist of three parts: a 5-carbon sugar, a phosphate group, and a nitrogenous base. Nucleic acids store and transmit bereditary, or genetic, information. There are two kinds of nucleic acids: ribonucleic acid ( RNA) and deoxyribonucleic acid ( DNA) Proteins are macromolecules that contain nitrogen as well as carbon, hydrogen, and oxygen. Proteins are polymers of molecules called amino acids. Some proteins control the rate of reactions and regulate cell processes. Some are used to form bones and muscles. Other transport substances into or out of cells or help to fight disease. |
2.4 Chemical reactions and enzymes
|
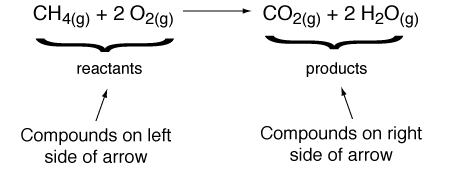
Chemical reactions is a process that changes, or transforms, one set of chemicals into another.
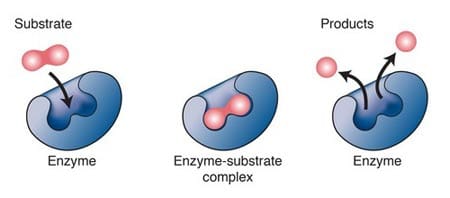
Reactants are the elements or compound that enter into a chemical reaction. Products are the elements or compounds produced by a chemical reaction. Chemical reactions always involve changes in the chemical bonds that join atoms in compounds. Some chemical reactions release energy, and other reactions absorb energy. Energy changes is an important factors in determining whether a chemical reaction will ocur. Chemical reactions that release energy often occur spontanously. Chemical reactions that absorb energy will not occur without a source of energy. Activation energy is the energy that is needed to get a reaction started. Catalyst is a substance that speeds up the rate of a chemical reaction, it works by lowerig a reaction's activation energy. Enzymes are proteins that act as a biological catalysts. Enzymes speed up chemical reactions that take place in cells. Substrates are the reactants of enzyme-catalyzed reactions. |
http://www.grandinetti.org/chemical-reactions
http://www.rethinkingcancer.org/resources/articles/what-do-you-know-about-enzymes.php
|